SnapDragon
Product Brief
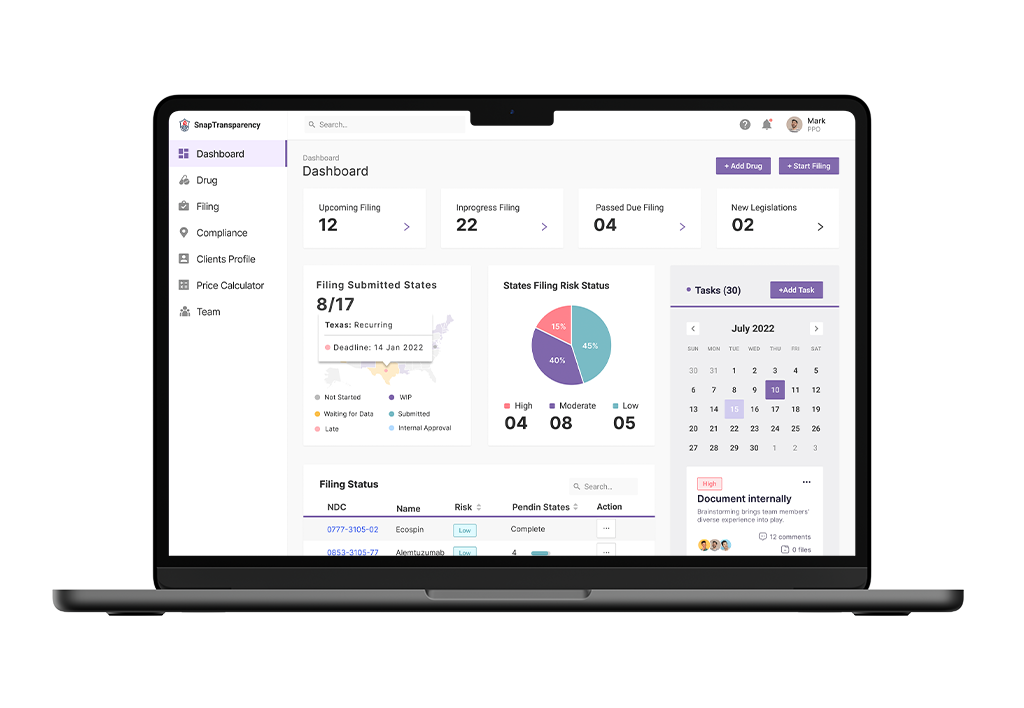
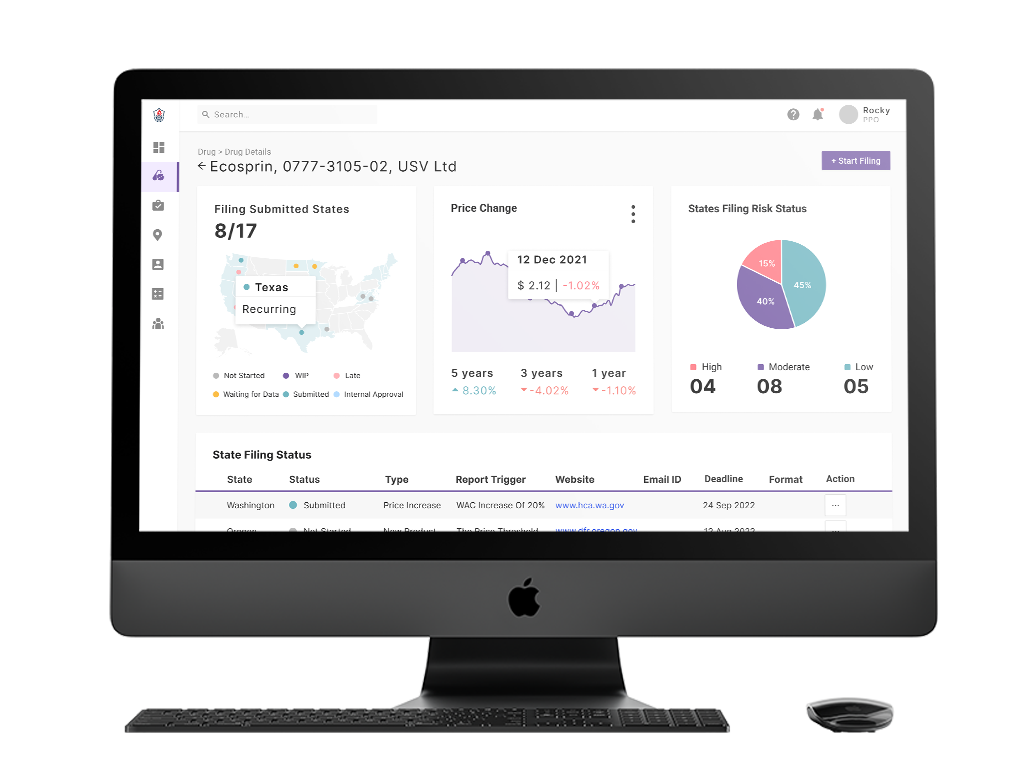
The Price Transparency product - Snap Transparency - makes price transparency compliance easier for Pharma companies.
It collects all the relevant information from the rulings and regulations across various states in the US and identifies compliance mandates for a given drug, for initial launch or subsequent price changes, what needs to be done in which state, and by when.
It automates compliance filings across every state by collecting, formatting, and communicating in the desired formats after applying all the relevant rules and calculations, thereby drastically reducing the (error prone) manual effort that goes into creating, reviewing and authorizing compliance submissions.
Missing compliance mandates and deadlines often attract heavy penalties, computed per day, per drug code. This not only delays time to market but increases governance overhead over time. Automation of compliance mandates secures this risk.
The Start
The Client and its founding team have a rich professional history in Pharma Industry. They needed a team that could take the idea and makes it real without being too dependent on their time since the product was being created in stealth mode. They also wished to build a team to ramp up further to create two other products in the same business domain. The Client wanted to focus primarily on sales and customer success, collaborate on the product roadmap and features and delegate the rest to Caizin. The Client was keen to hire a few team members from Caizin and make them part of their organization at a future date based on the product's success in the marketplace.
First 30 days of our engagement
We understood the initial product vision from the Client through initial meetings. We sponged the domain through our research and the client's critical resources. We documented our research into Confluence with ample details. We interviewed Client SMEs, a handful of end users, and industry leaders to understand how compliance is done today, what tools/alternatives are utilized, and the most challenging parts of the complete lifecycle. We created the re-stated problem statement, the value proposition design, and journey maps of 4 prominent user personas. We reviewed the problem statement with the stakeholder and confirmed our more profound understanding of the problem and intended solution. We re-interviewed the users in each user persona and validated the journey map to narrow down on critical challenges and critical benefits the product shall address. That is how the solution differentiator was uncovered. We assisted in creating an initial business and go-to-market strategy through the Lean Canvas exercise. In the first 30 days, we created a detailed roadmap and product MVP release plan and seeded the product team with key team members.
Our Collective Success
In the first six months, we completed the go-to-market version of the product.
From months three to six, we assisted and took executive ownership of launching the product marketing website.
Our Learning
We realized that for a high net worth market segment like Pharma, the approach towards keeping the features to a minimum in the MVP may not always work. The Customers appreciate it if all the features are in the first version since the replacement cost / adopting a new alternative is very high.
We realized that the buyers might have more motivation than just the features in an industry where technology change is costly. The visual appeal of the solution, more assistance in launch than just being self-serve, and ancillary decision-making data/information - they all influence the sales cycle.
We realized that treating the product program as a project for stealth mode startups makes sense since the budget is very discreet, and future budgets depend on the success of the first launch milestone. Sustainability is more important than continuity.